Have you ever wondered why iron rusts, why batteries give us power, or how our body uses food to produce energy? Behind all of these processes lies a very important concept in chemistry: redox reaction definition and example.
Redox reactions are one of the most fundamental topics in chemistry. They may sound complex at first, but once you understand the basics, you’ll see that they are happening all around us, every single day. From the air we breathe to the fuel that powers vehicles, redox reactions play a key role in life and technology.
In this blog, we will simplify redox reaction definition and example, break down the meaning of oxidation and reduction, and give you easy examples that connect chemistry to real life.
By the end, you’ll not only understand the redox reaction definition and example but also realize why this concept is so powerful in science.
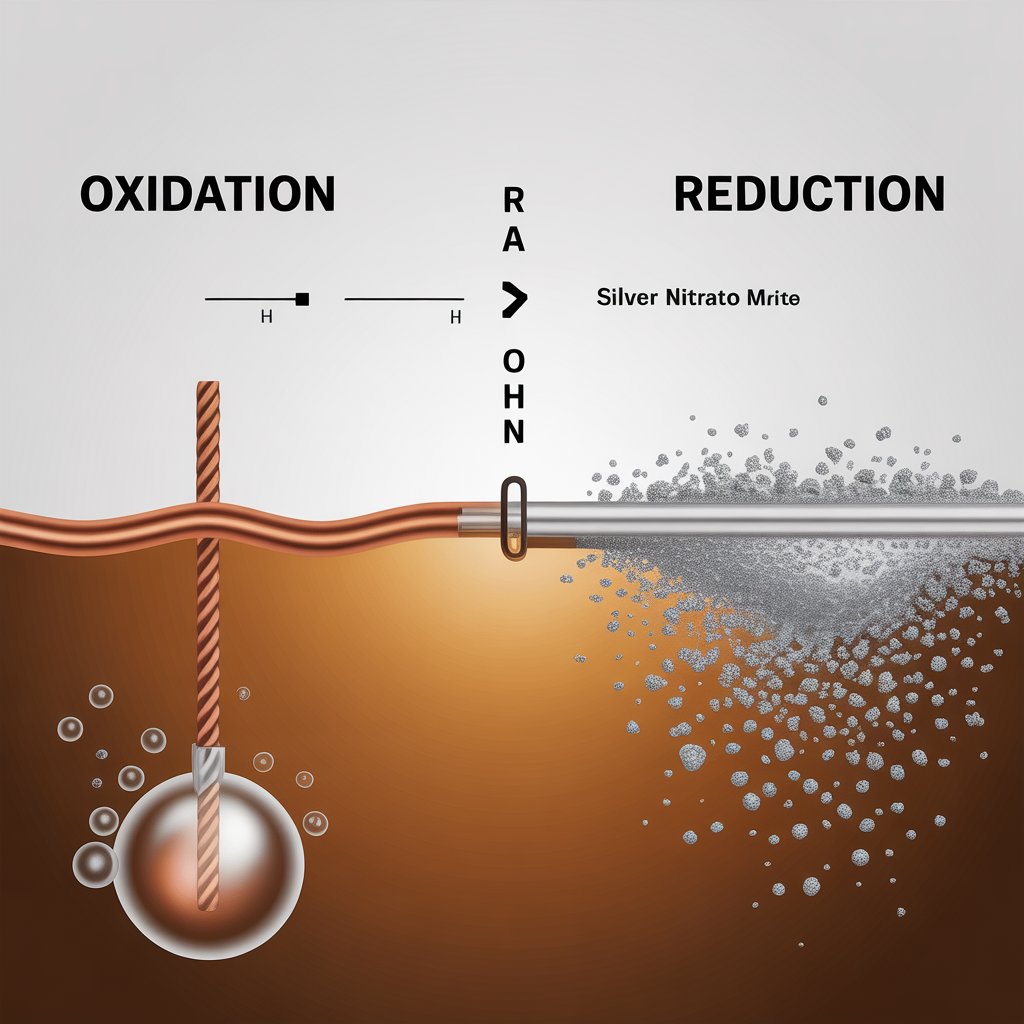
What is a Redox Reaction?
Many chemical reactions involve the transfer of electrons from one chemical substance to another. These electron transfer reactions are termed oxidation-reduction, or Redox reactions, which involve oxidation and reduction simultaneously. Redox reaction can also be termed as an increase in oxidation number or a decrease in oxidation number simultaneously.
Oxidation and Reduction Explained
To truly understand redox reactions, let’s break down oxidation and reduction in the simplest way possible.
🔴 Oxidation
- Loss of electrons
- Gain of oxygen
- Loss of hydrogen
- Increase in oxidation number
👉 Example: When magnesium burns in oxygen: 2Mg+O2→2MgO2Mg + O₂ → 2MgO2Mg+O2→2MgO
Here, magnesium gets oxidized because it loses electrons to oxygen.
🟢 Reduction
- Gain of electrons
- Loss of oxygen
- Gain of hydrogen
- Decrease in oxidation number
👉 Example: In the reaction of copper oxide with hydrogen: CuO+H2→Cu+H2OCuO + H₂ → Cu + H₂OCuO+H2→Cu+H2O
Here, copper oxide is reduced to copper because it loses oxygen.
✨ Quick Table for Clarity
| Process | Definition | Example |
|---|---|---|
| Oxidation | Loss of electrons / gain of oxygen | Mg → Mg²⁺ + 2e⁻ |
| Reduction | Gain of electrons / loss of oxygen | Cu²⁺ + 2e⁻ → Cu |
How to Identify a Redox Reaction
Many students find it tricky to spot a redox reaction in equations. Here are simple ways:
- Check for Electron Transfer – If electrons are moving from one element to another, it’s redox.
- Look for Oxidation Number Changes – If the oxidation number of any element increases (oxidation) and decreases (reduction) at the same time, it’s redox.
- Observe Oxygen or Hydrogen – Gain/loss of oxygen or hydrogen often signals redox.

Oxidising Agent or Oxidants
The substances that accept electrons in a chemical reaction, i.e., electron acceptors, are oxidising agents:
- Oxidising agents are Lewis acids.
- Substance which can oxidise others and reduce themselves.
- Substance which shows decrement in oxidation number.
- All high electronegative elements like N, O, F, Cl, etc are oxidants.
- All metallic oxides are oxidants.
- Some non metallic oxides are also oxidants like CO2, CaO, etc.
Reducing Agents or Reductant
The substances which donate electrons in a chemical reaction are reducing agents i.e electron donors are reducing agents.
- Reducing agents are Lewis base.
- Substances which can reduce others and oxidise themselves.
- Substance which shows decrement in oxidation number.
- All metal like K, Mg, Ca, etc are examples of reductants.
- All metallic hydrides are examples of reductants.
- All hydro acids are reductants. Example: HF, HCl, HBr, etc.
Redox Reaction Examples
Now let’s explore some examples to make this concept crystal clear.
1. Reaction of Zinc and Copper Sulphate
Zn+CuSO4→ZnSO4+CuZn + CuSO₄ → ZnSO₄ + CuZn+CuSO4→ZnSO4+Cu
- Zinc (Zn) loses electrons → oxidized
- Copper ions (Cu²⁺) gain electrons → reduced
This is a classic redox reaction taught in schools.
2. Rusting of Iron
Rusting is a redox reaction that occurs slowly in the presence of water and oxygen. 4Fe+6H2O+3O2→4Fe(OH)34Fe + 6H₂O + 3O₂ → 4Fe(OH)₃4Fe+6H2O+3O2→4Fe(OH)3
- Iron is oxidized to form Fe²⁺ and Fe³⁺ ions.
- Oxygen is reduced.
That reddish-brown rust you see on old iron objects? That’s redox in action.
3. Respiration in Living Organisms
When we breathe in oxygen and break down glucose, it’s a biological redox reaction. C6H12O6+6O2→6CO2+6H2O+EnergyC₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + EnergyC6H12O6+6O2→6CO2+6H2O+Energy
- Glucose is oxidized.
- Oxygen is reduced to water.
Without redox reactions, life would not exist!
4. Photosynthesis in Plants
Plants use sunlight to carry out the reverse process of respiration. 6CO2+6H2O→C6H12O6+6O26CO₂ + 6H₂O → C₆H₁₂O₆ + 6O₂6CO2+6H2O→C6H12O6+6O2
- Water is oxidized to release oxygen.
- Carbon dioxide is reduced to form glucose.
Photosynthesis is a life-sustaining redox process for our planet.
5. Batteries and Electrochemical Cells
Batteries that power our devices are based on redox reactions.
- At the anode, oxidation occurs.
- At the cathode, reduction occurs.
This flow of electrons generates electricity.
✅ Conclusion
Redox reactions may look complicated at first glance, but once you break them down into oxidation and reduction, everything becomes simple. They are the heartbeat of chemistry—present in our bodies, the environment, and the technology we use every day.
Mastering this topic is not just about exams but about understanding the invisible chemistry of life itself.